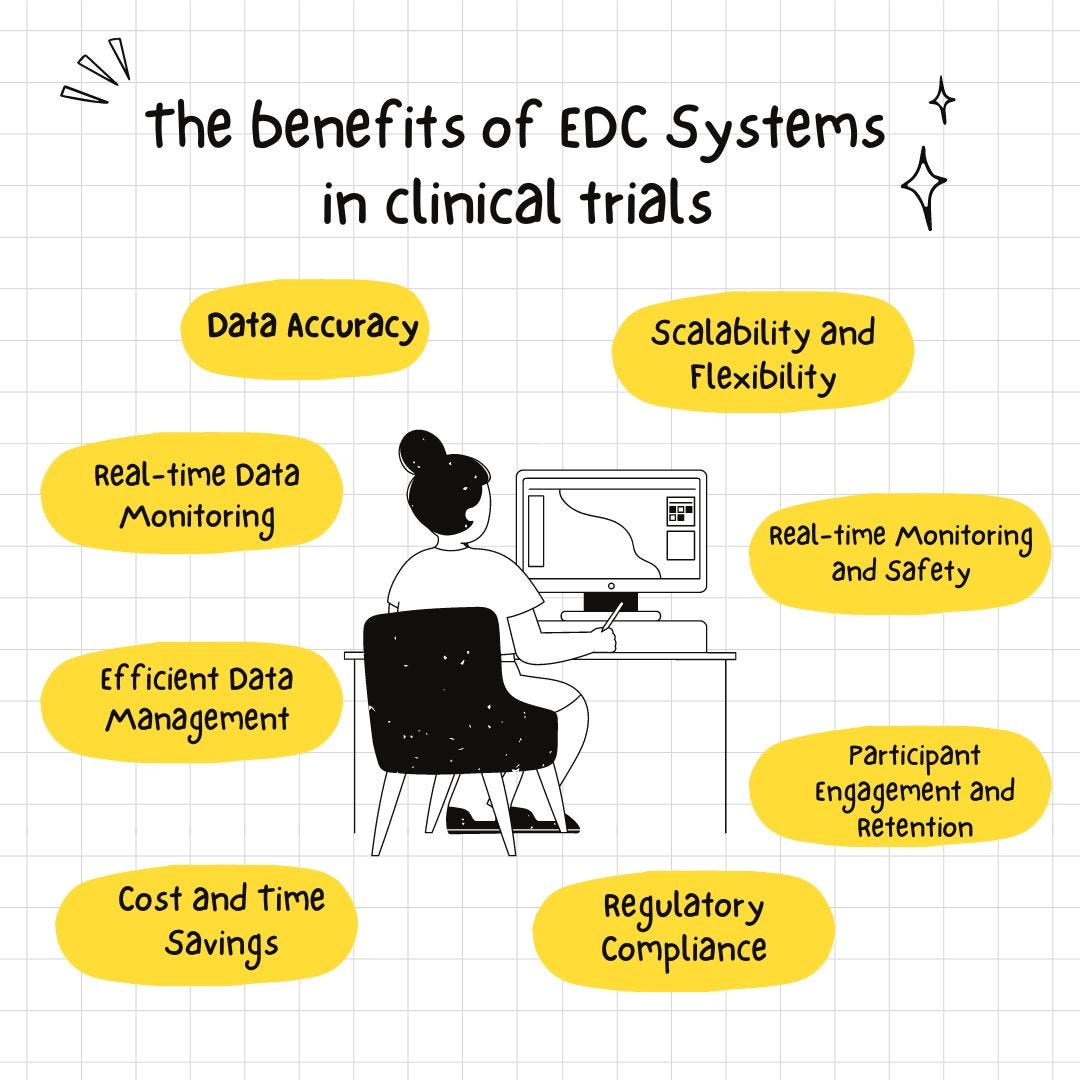
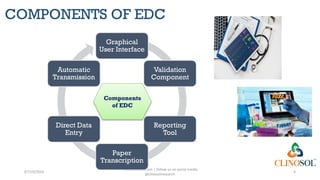
Figure 2 from Mobile Device–Based Electronic Data Capture System Used in a Clinical Randomized Controlled Trial: Advantages and Challenges | Semantic Scholar
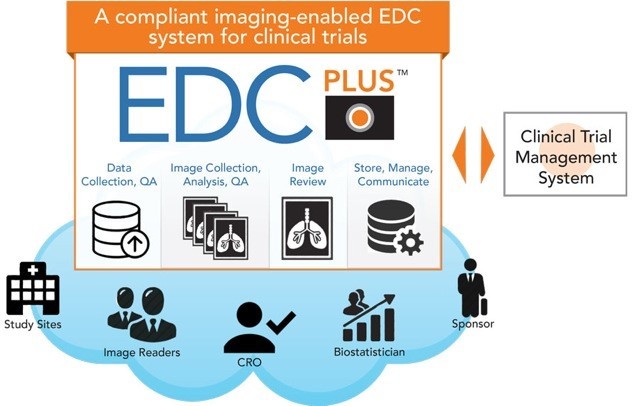
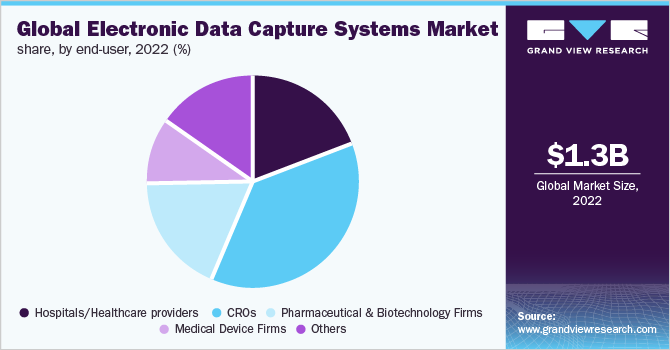
ImageIQ Releases EDCplus Technology, an Imaging-enabled Electronic Data Capture System for Clinical Trials | Business Wire
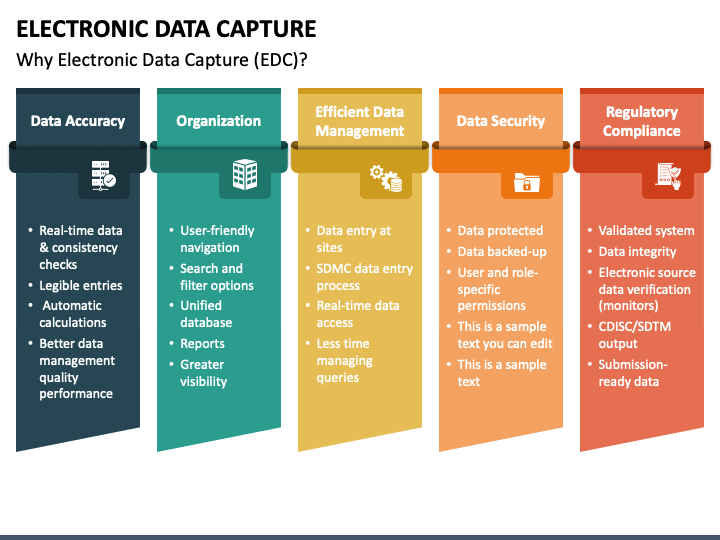
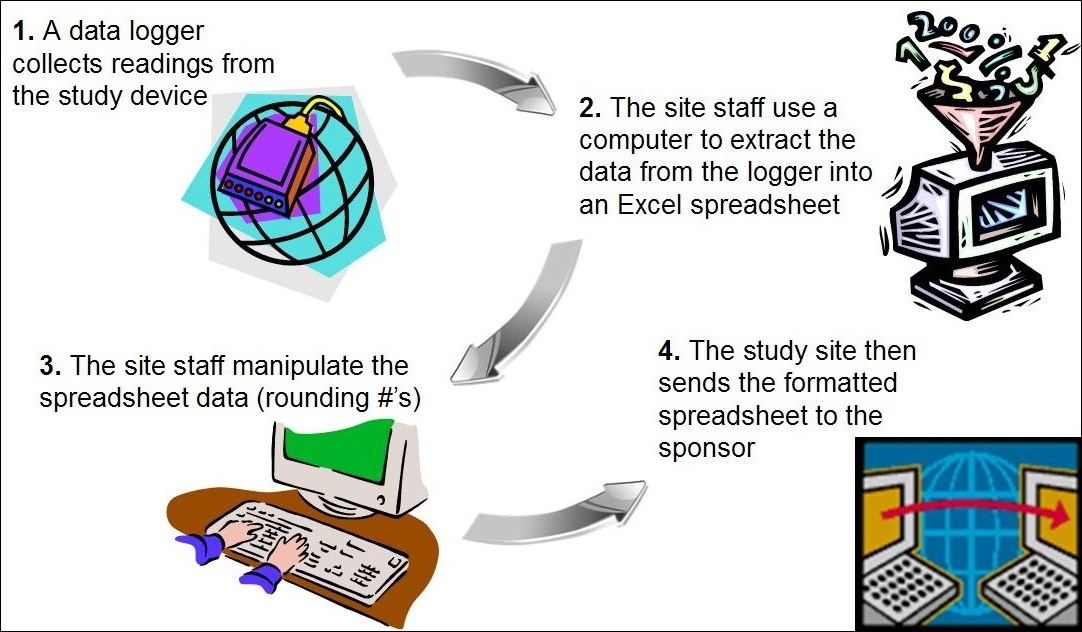
![PDF] Electronic Data Capture in clinical trials– interface design and evaluation and system validation | Semantic Scholar PDF] Electronic Data Capture in clinical trials– interface design and evaluation and system validation | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/50a3193b5f8fb7f91479fd8839c1a6d03baba9b1/19-Figure2-1.png)





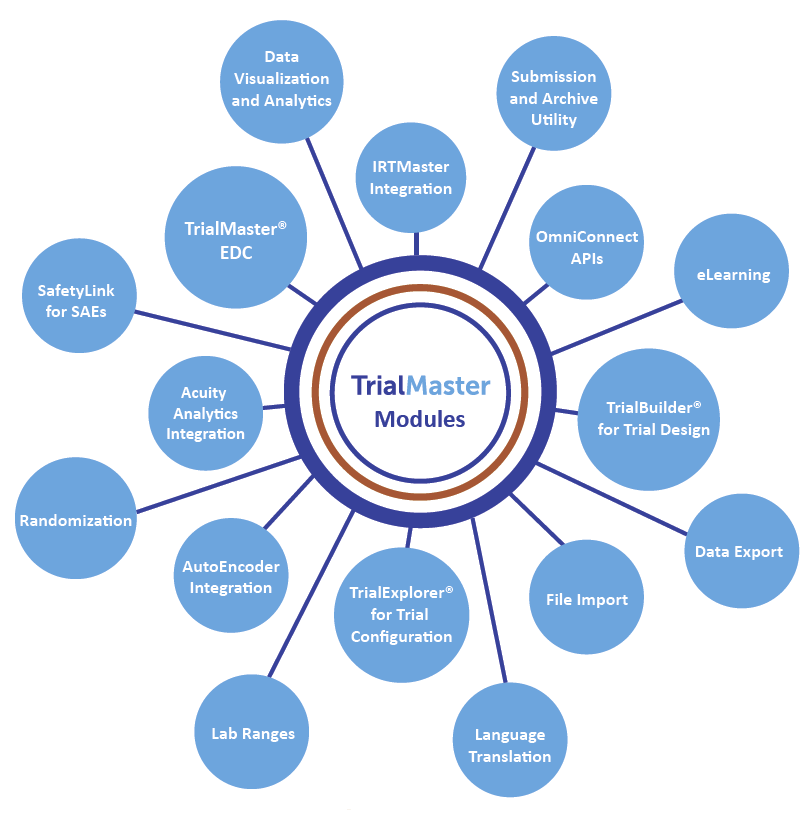
%20Systems%20(new).png?width=800&height=400&name=Everything%20You%20Need%20to%20Know%20about%20Electronic%20Data%20Capture%20(EDC)%20Systems%20(new).png)